Navigating the Integration Challenges of Automation in Drug Discovery: End-to-End Automated Synthesis

Integrating hardware and software to achieve automation in drug discovery processes, especially in chemical synthesis, is increasingly becoming the standard, revolutionizing the way scientists conduct their research. However, these innovations bring challenges such as data management, hardware interoperability, software compatibility, and process standardization.
To navigate these complexities, it is crucial for companies to adopt a holistic approach to their lab operations. Identifying the most suitable tools and leveraging existing resources are key steps in this process. By focusing on utilizing ready-made solutions and only building what is essential, organizations can streamline their workflows, enhance efficiency, and drive innovation in their drug discovery endeavors.
Integration Challenges

Software Compatibility and User Experience
The drug discovery process involves multiple stages, each relying on various software tools such as cheminformatics or bioinformatics tools, compound management and inventory software, HTS data analysis, ELNs, and LIMs. Ensuring seamless communication and data sharing between these systems is challenging due to compatibility issues arising from different data formats, proprietary systems, or outdated software. This can lead to inefficiencies and errors in the workflow.
In addition, software that isn’t user-friendly can frustrate end-users and scientists, resulting in poor adoption, data loss, and errors.
Hardware Interoperability
Labs often have several brands of hardware that need to be integrated within a lab in an automated workflow. These different robotics may have varying communication protocols, interfaces, and operating requirements, making seamless integration challenging. The result can be manual interventions, reduced throughput, and increased potential for errors.
Data Management
Data management includes several areas. One hurdle in data management is data silos where information is isolated and inaccessible to other parts of the organization, making it hard to leverage experiment data across groups and collaborate. This can also lead to redundancy and ineffective decision making due to incomplete data.
Standardizing data is important to ensuring various sources can be integrated and interpreted consistently. Standardizing formats, naming conventions, and metadata is critical to be able to combine and analyze this information effectively. Ideally, this is an organization-wide standardization that establishes common data models, controlled vocabularies, and data exchange formats.
Data cleaning is another essential part of data management, where data quality directly impacts research outcomes. This process gives you the confidence in your data integrity through tasks such as removing duplicates, standardizing data formats, imputing missing values, etc.
The data integration itself is particularly challenging because of the complexity and variety of data involved. To do this effectively, data solutions must be able to handle large volumes of data that is correctly aligned, merged, and harmonized. This is the best way to ensure a comprehensive view of the drug discovery process and to facilitate better analysis, visualization, and decision-making.
Process Standardization
Process standardization can be challenging due to the diversity of protocols, methodologies, and equipment used in different stages of the workflow. However, this is essential for consistency and reproducibility in drug discovery. Teams across the organization should follow the same procedures and equipment is calibrated, operated, and maintained in a consistent manner.
Overcoming Integration Challenges

Scientist-First, User-Friendly Software
Good software is software that is used consistently and with ease. This means using software that is intuitive, is tailored to your specific experimental setups and works with the hardware and software you want to integrate together, and adapts to scientists’ needs, not the other way around.
Modular, Vendor Agnostic Systems
Investing in architecting a modular hardware and software solution allows organizations to easily reconfigure their systems to accommodate new technologies or changes in workflow. Modular systems are meant to be interoperable, so you can easily change components without disrupting your entire workflow. This enhances integration as well as reduces time and cost associated with upgrading or scaling the system.
Leverage the Cloud
Centralizing data on cloud-based platforms can facilitate seamless integration across various stages of research to enable collaboration, access, and analysis. This allows scientist more flexibility in compute power, especially for large datasets, preventing bottlenecks. Cloud solutions also enhance data security with robust encryption and access controls.
Adopting Open Standards
A key strategy to overcome integration challenges is enhancing communication and collaboration with vendors to improve their APIs and drivers. Engaging with vendors allows organizations to provide feedback and insights into their drug discovery needs. This collaborative approach can lead to more flexible and compatible APIs and drivers, tailored to the scientific community's requirements. By working closely with vendors, organizations can ensure their tools are designed for interoperability, reducing compatibility issues and streamlining integration.
Selecting the Right Balance of Ready-Made and Made-for-You
A critical aspect of overcoming integration challenges in automation is selecting the right mix of standardized and customizable tools. The goal is to achieve a balance between ready-made solutions that offer essential features and scalability, and tailor-made solutions that cater to specific needs. Utilizing software that only partially meets your requirements, whether it's 20% or even 80%, can lead to inefficiencies and fragmented workflows. The key is to identify solutions that provide a solid foundation of core functionalities while also offering the flexibility to adapt and integrate with other systems as needed. This approach ensures a more seamless and effective integration process, ultimately enhancing the overall efficiency of your automation efforts.
Sample Solution: Project JedAI

At Integrant, we believe in leveraging what you can and building only what you need. Our sample solution, Project JedAI, was designed with compound synthesis in mind but can be applied to biologics or other areas of drug discovery where automation integration is desired. It serves as a wrapper for integrated systems, allowing you to utilize all your systems in one place.
Project JedAI is user-friendly and integrates key systems of your choice, showcasing the possibilities for data management, software compatibility, and hardware interoperability. It uses ChemAxon tools such as JChem Microservices DB, Microservices Calculations, Microservices Reactor, and MarvinJS, and can integrate with LIMS, sample management, ELN, and other software like Mosaic, Benchling, GBG, and more.
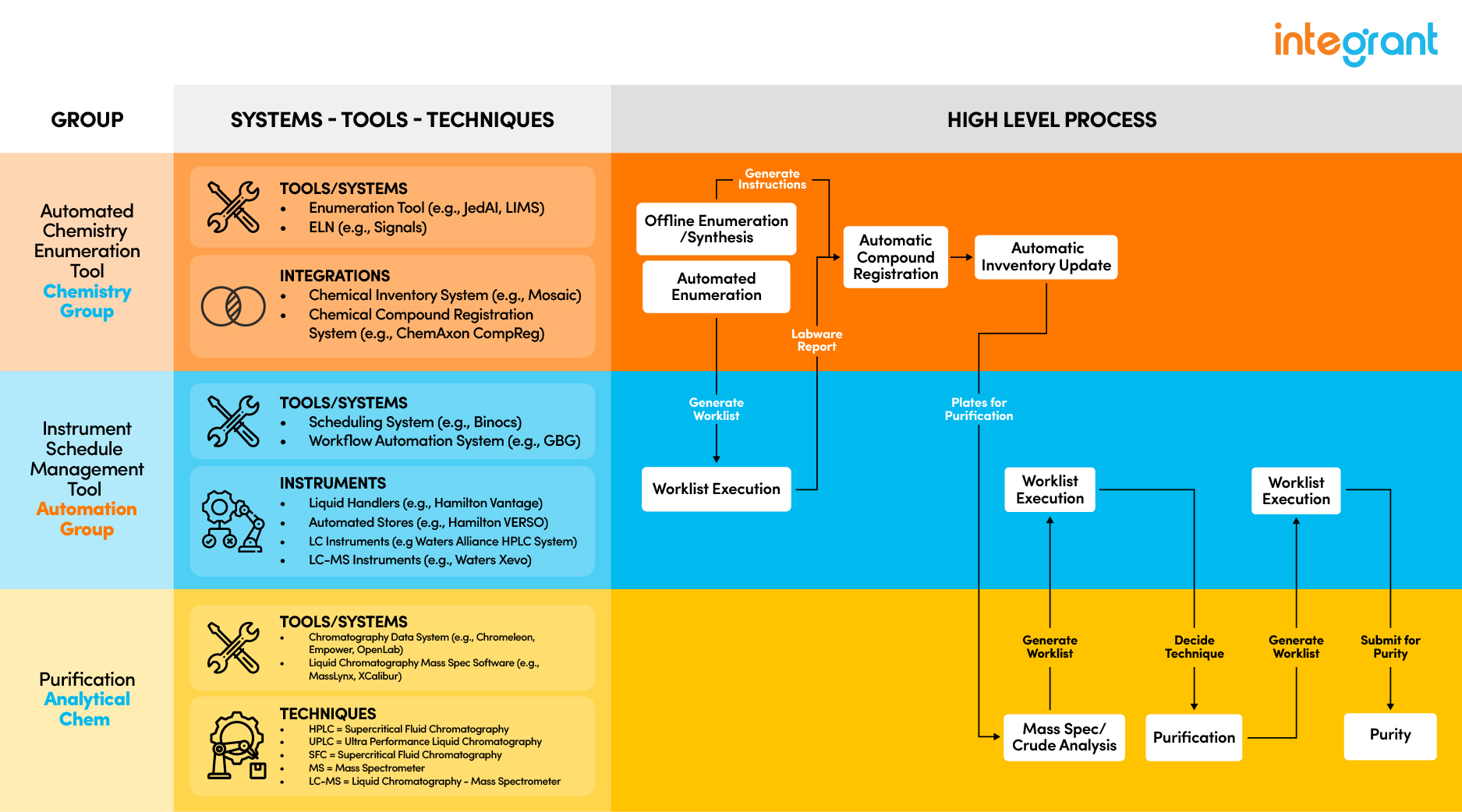
Above is a sample diagram of one workflow that can be implemented via JedAI. You can customize any workflow and steps to match your group and organization's needs.
Below is a sample diagram of one workflow that can be implemented via JedAI. You can customize any workflow and steps to match your group and organizations’ needs.
For a closer look at Project JedAI and how this can streamline your integration and automation, schedule a meeting with our team today.